When an innovative therapy aiming for a "one-time cure" is still evaluated by traditional standards that measure "months of survival," its failure may have been predetermined. In 2025, setbacks in several high-profile CGT clinical trials served as a resounding wake-up call for the entire industry. This signals that a disruptive revolution in the clinical evaluation system—from "treatment" to "cure"—is already on the horizon.
Cell and Gene Therapy (CGT) has become a core growth engine in the global biopharmaceutical sector. Authoritative forecasts predict that the global CGT market will climb to $30.54 billion in 2025, with a Compound Annual Growth Rate (CAGR) as high as 65.8% from 2023 to 2025.
China's performance in this field is equally remarkable. As of the second quarter of 2025, the Center for Drug Evaluation (CDE) of the National Medical Products Administration (NMPA) had accepted 765 Investigational New Drug (IND) applications for CGT products, demonstrating strong R&D momentum.
However, behind the frenzy of capital and the expansion of drug pipelines, the CGT industry in 2025 is experiencing profound divergence and growing pains. Major global regulatory agencies continue to tighten their review standards for CGT products, exposing weaknesses such as immature technology and insufficient preclinical research.
A series of events, including the suspension of Phase III clinical trials and the rejection of Biologics License Applications (BLAs), indicate that CGT R&D has entered a "deep-water zone." Technological breakthroughs alone are no longer sufficient to ensure product success. Scientifically rigorous trial design and full lifecycle data management capabilities have become critical determinants of success, compelling the CGT clinical evaluation system to undergo a fundamental reconstruction.
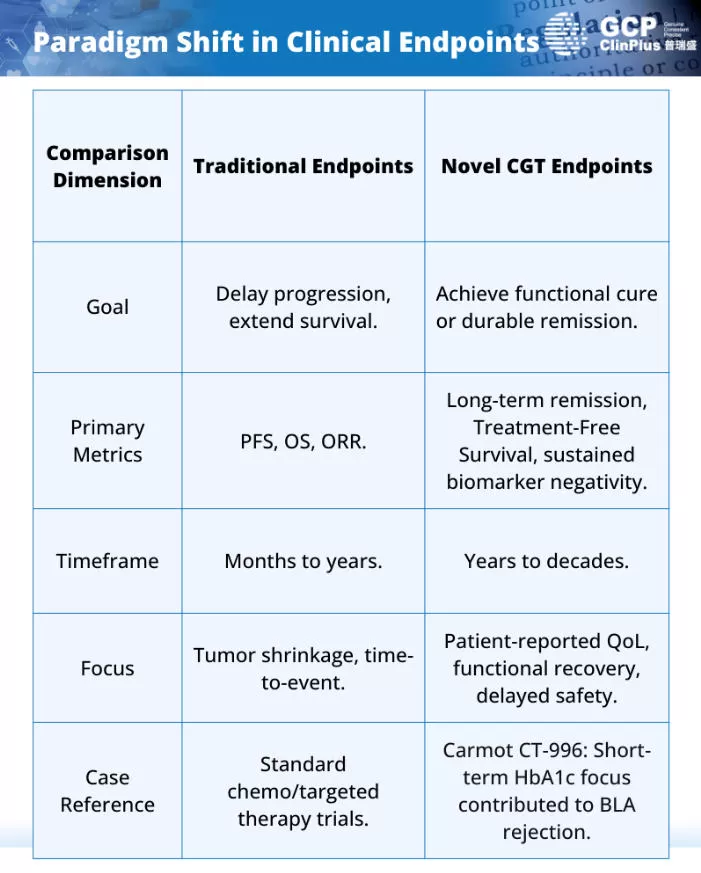
The industry's collective challenges point to a core issue: there is a fundamental mismatch between the "cure" objective of CGT therapies and the "delay" mindset inherent in traditional clinical endpoints. This mismatch necessitates a comprehensive upgrade of the CGT clinical evaluation system, from conceptual understanding to practical application.
CGT therapies, particularly cutting-edge technologies like CAR-T and gene editing, have an ultimate goal far beyond simply controlling disease progression; it is to achieve functional cures or long-term remission. This implies that the evaluation logic of clinical trials requires a fundamental shift. Traditional oncology endpoints such as Progression-Free Survival (PFS) and Overall Survival (OS) appear narrow and inadequate when applied to CGT therapies pursuing a "one-time cure."
CGT clinical trials demand novel endpoint designs, with the core focus on demonstrating patients' "long-term benefit":
Long-term Remission Rate: Emphasizes the duration of sustained remission after treatment, directly addressing the core demand for a "cure."
Treatment-Free Survival (TFS): Measures survival time without the need for subsequent therapies, reflecting the long-lasting value of the treatment.
Sustained Negativity of Biomarkers: For example, sustained negativity of Minimal Residual Disease (MRD) after CAR-T therapy is a key indicator for evaluating curative potential.
Patient-Reported Outcomes (PROs) and Quality of Life (QoL): The meaning of a cure encompasses not only biological remission but also the restoration of patients' physiological and social functions.
Industry Warning: Endpoint designs adhering to traditional thinking have become a significant cause of CGT product failures. For instance, in 2025, the primary endpoint for the Phase III trial of Carmot Therapeutics' innovative therapy CT-996 for type 2 diabetes was set as the reduction in glycated hemoglobin (HbA1c) levels. In the context of the FDA's heightened focus on the clinical value of innovative therapies, this design—solely focused on short-term glycemic control—failed to meet stringent approval standards. Consequently, the BLA for this product was rejected. This case clearly warns that CGT clinical trial endpoint design must move beyond the limitations of short-term indicators. It must integrate long-term efficacy, safety, and improvements in patient quality of life into its core considerations, using forward-looking scientific design to match the therapy's "cure" potential with regulatory scientific requirements.
Cure-oriented endpoint design imposes higher demands on clinical trial execution, with the foremost challenge being ultra-long-cycle follow-up work. Unlike the short-term observation required for traditional therapies, the unique risk profile of CGT therapies makes ultra-long-term follow-up an essential path to verifying "cure" effects.
The potential risks of CGT therapies—such as delayed adverse reactions, genomic integration risks, and long-term immune memory issues—dictate that clinical trial follow-up cannot end at the traditional "study conclusion." Currently, major global regulators like the FDA, EMA, and NMPA have clearly mandated that CGT trials must establish Long-Term Follow-Up (LTFU) plans, often spanning 10 to 15 years. For pediatric patients, the follow-up period may even need to extend into adulthood.
Multiple failure cases in 2025, at a heavy cost, have confirmed the extreme necessity of ultra-long, high-quality follow-up and revealed three core challenges behind it:
Challenge 1: Insufficient Follow-up Duration, Leading to Regulatory Scrutiny of Efficacy Evidence
In July 2025, Capricor Therapeutics' cell therapy Deramiocel (CAP-1002) was denied approval by the FDA. One of the main reasons was deficiencies in the key clinical trial data. The study not only had a small sample size but also a follow-up period of only 6 months. For a progressive disease like Duchenne Muscular Dystrophy (DMD), such short-term data is completely inadequate to assess the therapy's long-term impact on disease progression, naturally failing to meet the stringent regulatory requirements for "substantial evidence." This warns the industry: the duration of CGT follow-up plans must align with the natural history of the disease and the theoretical benefit period of the treatment.
Challenge 2: Lagging Safety Monitoring, Making It Difficult to Capture Delayed Toxicities
Toxic reactions to CGT therapies are often delayed and unpredictable, rendering the short-term safety observation windows used for traditional drugs entirely obsolete. In 2025, Intellia Therapeutics' CRISPR gene-editing therapy nex-z was urgently paused in its Phase III trial after a patient developed severe delayed hepatotoxicity meeting "Hy's law" (Grade 4 elevated liver enzymes accompanied by bilirubin rise) more than three weeks post-treatment. Similarly, Fosun Kite's CAR-T therapy FKC889 was forced to halt clinical advancement due to subjects experiencing severe neurotoxicity (Grade 4 encephalopathy). These cases fully demonstrate that CGT clinical trials must establish active monitoring systems covering several years or longer to effectively capture rare delayed adverse events.
Challenge 3: Data Management and Patient Retention: A Decade-Spanning Practical Dilemma
A follow-up cycle exceeding ten years inevitably faces the persistent challenge of high patient dropout rates. Furthermore, unifying data standards across different research centers and time periods is exceptionally difficult. To solve this problem, it is necessary to abandon traditional manual follow-up models and adopt digital, patient-friendly follow-up strategies.
The "impossible triangle" of ultra-long-term follow-up has made traditional follow-up models unsustainable. To break this deadlock, the industry is beginning to explore new solutions centered on digitalization and ecosystem integration, driving the CGT clinical evaluation system toward greater efficiency and sustainability.
Faced with the systemic challenges of CGT ultra-long-term follow-up, reliance solely on traditional manual follow-up is no longer viable. The industry is currently exploring the construction of a sustainable follow-up ecosystem based on real-world data, decentralized models, and artificial intelligence (AI) technology to break through execution bottlenecks via technological innovation.
Decentralized Follow-Up: Utilizing a "virtual research site" model that integrates telemedicine, wearable devices, and electronic Patient-Reported Outcome (ePRO) tools is a key strategy to minimize patient follow-up burden and maintain participation in ultra-long-term studies.
Federated Data Networks: Establishing flexible data collaboration networks that allow data to be analyzed collaboratively while remaining securely stored locally can break down data silos and promote efficient, compliant collaboration among pharmaceutical companies, hospitals, and regulators in long-term follow-up.
Intelligent Risk Prediction: Leveraging AI algorithms to deeply analyze longitudinally accumulated clinical data can accurately predict patient dropout risks or potential safety events, enabling personalized proactive management and transforming patients from passive subjects into active partners.
Amid the industry's active exploration of new paradigms, GCP ClinPlus leverages its profound project experience and self-developed intelligent platform to translate the concept of evaluation system innovation into actionable solutions, providing full-chain support for the "cure" journey of CGT therapies.
As a pioneer in the domestic CGT clinical trial field, GCP ClinPlus has cumulatively advanced over 80 related studies, accumulating deep project experience. Relying on its self-developed clinical research intelligence platform, Clinx, GCP ClinPlus transforms industry insights into practical solutions, providing full-chain intelligent support for CGT R&D.
Scientific Endpoint and Statistical Strategy: Deeply assists sponsors in formulating scientific, forward-looking study protocols and statistical plans, ensuring trial design reflects the therapy's "cure" potential while precisely meeting global regulators' stringent requirements for long-term evidence.
Intelligent Follow-Up and Data Management: Addressing the pain points of ultra-long-term follow-up, our system supports automated, cross-site task management and data synchronization, and actively explores integrating remote follow-up tools to enhance patient retention and data continuity.
Proactive Risk Monitoring and Quality Control: Utilizes AI models for real-time analysis of project progress and safety data, enabling proactive risk warnings and building a more sensitive risk control system than traditional models.
Professional Global Regulatory Submission Support: Possesses a deep understanding of the increasingly stringent technical requirements for CGT products from regulators like the FDA, EMA, and NMPA. Provides end-to-end data management and submission services compliant with international standards, assisting products in efficiently passing review.
As the partner for the first approved stem cell product in China, GCP ClinPlus has not only accumulated full-cycle practical experience from R&D to market but also provides robust quality and process assurance for addressing complex challenges in CGT clinical operations.
The endpoint of a CGT clinical trial is never the "conclusion" of the research, but the "beginning" of long-term patient management.
From "treatment" to "cure," from "follow-up" to "lifetime management," this disruptive evolution of the clinical evaluation system not only reconstructs the endpoint design and follow-up models of clinical trials but also reshapes the entire R&D logic of the CGT industry. The industry lessons of 2025 have shown that only by deeply integrating solid technological foundations, scientific clinical design, stable manufacturing processes, and intelligent full-cycle management can we navigate R&D risks, truly unlock the curative potential of CGT, and translate medical hope into a tangible reality for patients.
Sources:
《2025-2029 China Cell and Gene Therapy (CGT) Industry Investment Planning and Prospect Forecast Report》
《Nature Reviews Drug Discovery》
《Trends in the development of cellular and gene therapy in China. Nature Reviews Drug Discovery. 2025.》
This article is written based on the extensive collection and systematic review of industry reports and online materials. The content is for learning, exchange, and industry reference only. We strive to provide accurate and complete information but cannot guarantee absolute infallibility and assume no responsibility for any direct or indirect losses arising from the use of this content. Matters not covered in this disclaimer are subject to relevant national laws and regulations.
Preview
On February 25, we will release "(CGT Topic - Part 2): How to Design Compliant Protocols for Innovative CGT Clinical Trials," focusing on China-US-EU regulatory differences and Pre-IND communication strategies. Stay tuned.